This is the seventh live-blog of my spring 2026 DERs class.
The last post introduced linear ordinary differential equations as a tool for DER modeling. Instead of leading with the general theory, as the lectures do, the last post led with a DER we’re all familiar with: The humble battery. It turns out that learning to model batteries teaches us almost everything we need to know to model a wide variety of other DERs, such as space heating and cooling systems, water heaters, thermal storage, and others.
This post will talk a bit more about batteries. Specifically lithium-ion batteries, which power almost all laptops, phones, and e-mobility, from wheelchairs to semi trucks. Lithium-ion batteries have high energy densities, charge and discharge quickly, and last a long time. Oh, and they’re now dirt cheap.
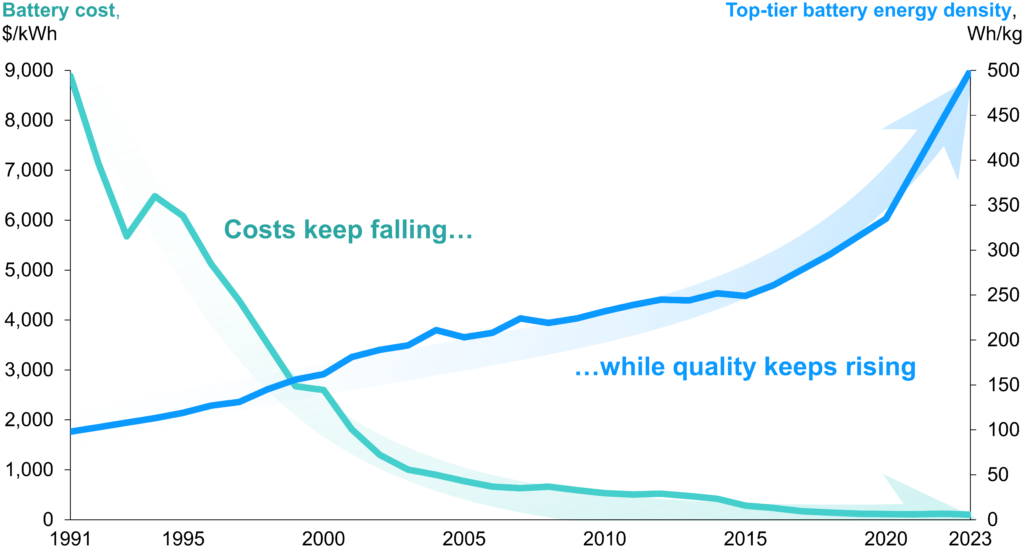
I’m not a chemist, so I’ll skip writing about how batteries work and refer interested readers to this nice article from the Australian Academy of Science. I also won’t write again about modeling how the energy stored in a battery evolves over time; the last post did that. Instead, I’ll focus on how to model energy conversion losses when charging or discharging. I’ll also talk a bit about how to pick reasonable values for the parameters in battery models.
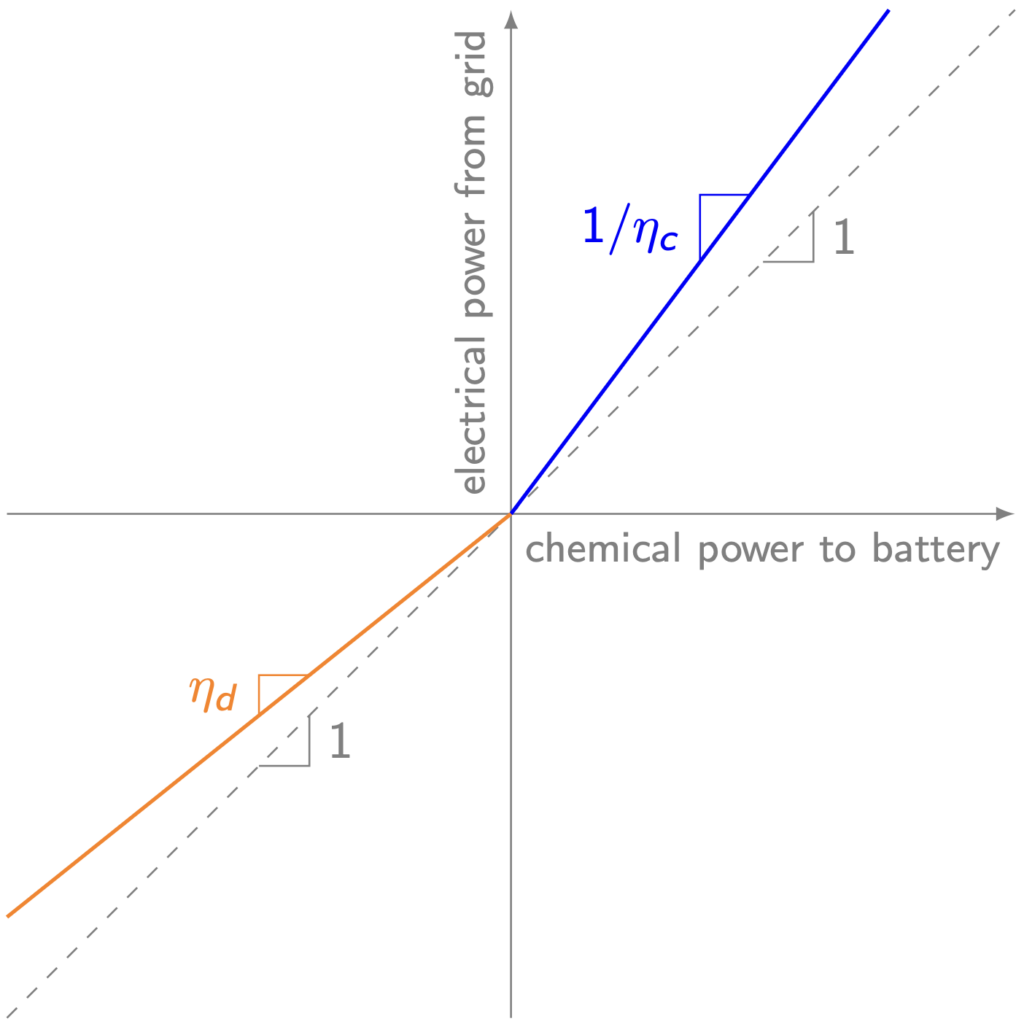
Devices that convert energy from one form to another generally lose some energy as waste heat. An electric motor, for example, might turn 90 percent or so of the electrical energy it draws from the power grid into rotational kinetic energy; the other 10 percent dissipates to the surroundings via heat transfer as friction and electric resistance heat up parts of the motor. Similarly, a battery might lose five percent or so of the input electrical energy when it charges and another five percent or so when it discharges. As shown below, the battery’s charging (blue) and discharging (orange) efficiencies determine the energy conversion losses.
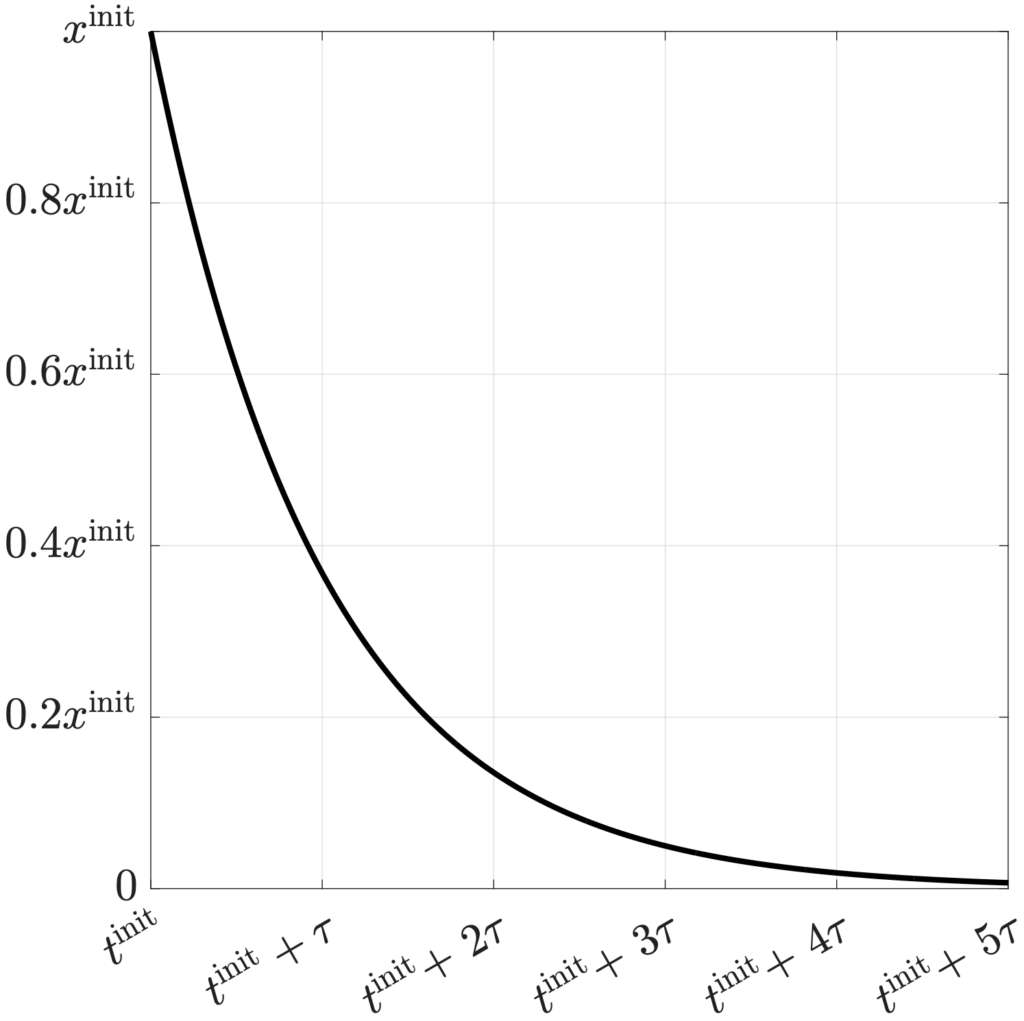
Along with the charging and discharging efficiencies, one more parameter governs a battery’s energy losses: The self-dissipation time constant. As discussed last time, self-dissipation is the slow, passive loss of energy to the surroundings that happens even when a battery is unplugged and unused. Self-dissipation is what drains a battery forgotten in a junk drawer. As with the charging and discharging efficiencies, higher values of the self-dissipation time constant are better. The figure below shows how an unused battery’s stored energy decreases with time. After three time constants, about 95% of the initial energy has been dissipated as heat. A typical lithium-ion battery has a time constant on the order of 1,000 hours.
Three more parameters are needed to fully define our battery model: The energy capacity (in kWh) and the charging and discharging power capacities (in kW). These are all design parameters that are typically determined by the application. For most lithium-ion batteries, the energy capacity is two to four hours times the charging power capacity. For stationary batteries, the discharging power capacity is typically similar to the charging power capacity. For electric vehicle batteries, the discharging power while traveling is, for all intents and purposes, unlimited. When you floor it in an electric car — to go from zero to 60 miles per hour in under two seconds, say — the battery discharges at over 500 kW. For context, Level 1 electric vehicle charging is around 2 kW. Level 2 is around 10 to 12 kW.